West coast volcanos/thermals hide GOLD!
I just saw a public tv show on minerals, gold, platinum just now and sat down to write this while it was fresh in my mind. Now that GOLD is at 1700$ an oz, it is TIME TO PAN gold, FIND GOLD, METAL DETECTOR UP SOME GOLD, (LOCATE PLACES/ SERVICES of every kind ( http://www.goldmaps.com/links.html ) or if You are rich, BUY GOLD. Read where, how, who and why by googling up reference pages, using words like PROSPECTOR, THERMAL, GOLD... you combine them or it doesn't work as well. MAPS. I'd add that. CAMPING and LODE. The show I was watching said that the nuggets always came up thru the smokestacks and/or thermals. They depicted sites in CYPRUS that were the antique copper/ gold mines that spawned the BRONZE age in 4000 BC. There were thermals in JAPAN rich in GOLD, and then they showed the under-sea mountain ridges that had these volcanic spouts that pulled minerals from deep below. And there was a picture of the undersea mountain ridge, how it crossed the PACIFIC and like a big, long DRAGON, threw itself across the CALIFORNIA, OREGON BORDER!I say we get a pick axe, hand sized, and hit the mts flanks where you see red iron deposits in the mountain walls as gold is there too. It is washed down hill by rain. A geologist told me: "There still is lots of Gold left behind in Calif. Any west-draining river off the Sierra Nevada Range has gold in it. The big problem is how much time do you have, how hard do you want to work and how much will you find. Panning is hard work, time-consuming and probably you will find some flakes here & there (not enough to pay the bills). In order to do better, you must build a sluice box and shovel lots of sand & gravel & dirt into it and let flowing water do the hard work for you. Or better yet, get a vacuum hose thing (like deep sea treasure hunters use) and suck up the sediment at the bottom of the stream beds and the stuff under boulders and run that thru the sluice box. It's a full time job and finding the sweet spots is trial & error."
What you want to do is find the DRAGON! THERE is an undersea crest in pacific, a mt top thingie, 46000 miles
long,winding like a snake, where it hits our westcoast is where these smoke stacks exude the minerals from earth's ctr. You want to find the thermal springs in that area, get a vacuum, sluice and pull the sand out from under rocks.
http://vulcan.wr.usgs.gov/Volcanoes/WesternUSA/Maps/map_west_coast_volcanoes.html
has a fine map showing volcanoes in this area.EXAMINE such sites, google these words "sea floor" + maps FIND the dragon's back where it winds ON TO DRY LAND Go there with pick, pan, lumber to make a sluice. tent, food, stove, light, a gun and a gal. A FRIDGE that locks so bears cannot smell ya.
http://www.ngdc.noaa.gov/mgg/image/2minsurface/45N135W.html
mining sourcesGoogle around looking for the sonoma-clear lake area of Northern California
GOOGLE or LOOK for a mix of smoke stacks, geo thermals, hot springs. Usually they're mentioned in TOURIST literature. "Hot Springs," like WHEELER SPRINGS tent out around that area. As GOLD is over 1300$ an oz!
~^~^~^~
NEXT, maybe we can envision a movie script written about this area, modern GOLD FEVER. A script which ultimately will give you a vacation in the area, say while doing the actual FILM SHOOT. We are there for three months. This area has history and elements that would make a flick. Say a historical western script,replete with California injuns who know where the hidden gold is. And some early white trash i.e. the villain. Set it in 1850's.
OR DO IT MODERN TIME.
BUT check out where THE DRAGON leaves the Pacific and crosses into the SIERRA NEVADA, I saw the best undersea map (better than those I've googled so far,) on the actual t.v show. DRAGON's back hits land somewhere right around Clear Lake. Volcanoes and smoke stacks and thermals characterise its passage across land. You wanna be there and vacation, tent there, pan there. Set scripts there. You know, maybe we could help each other on this? GOOGLE sonoma-clearlake + thermals+ panning + maps + hot springs
I GET THIS:
http://pubs.usgs.gov/of/1995/ofr-95-0831/CHAP25.pdf
http://www.youtube.com/watch?v=sCAka-w9X3I
http://www.metaldetector.com/learn/buying-guide-articles/gold-prospecting/whats-the-best-gold-prospecting-metal-detector
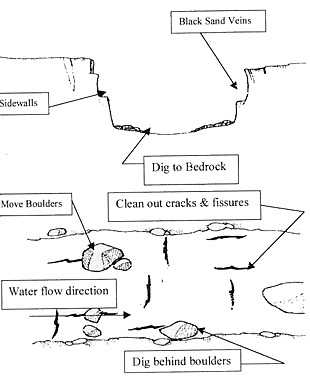
http://www.naturenw.org/rock-gold.htm mining instructions for amateurs.
it's always in quartz veins in place or pulled by water down into rivers.Thrust under rocks in curves.
Seek out a GOOGLE PAGE with a list of articles.
THE GOVERNMENT has pamphlets on this. The KEYWORDS to search on, prospecting, prospect gold, camping.
VIDEO on GOLD PLACER TECHNIQUES https://www.youtube.com/watch?v=UsJDEBpoNdU
VIDEO from a geologist prospector on WHAT ROCKS GOLD is found in: https://www.youtube.com/watch?v=e9rZxBvS1-Q
* * * * * * * * * * * * * * * * * * *<=== BACK TO THE FRUGAL TIPS and CHEAPO LIFESTYLE WEBPAGE
Our POSTER is ANITA SANDS HERNANDEZ, Los Angeles Writer, Frugal Wealth researcher single mother of 4 and career Astrologer . Catch up with her websites TRUTHS GOV WILL HIDE & NEVER TELL YOU, also The FUTURE, WHAT'S COMIN' AT YA! FRUGAL LIFE STYLE TIPS, HOW TO SURVIVE the COMING GREAT DEPRESSION, and Secrets of Nature, HOLISTIC, AFFORDABLE HEALING. Also ARTISANRY FOR EXPORT, EARN EUROS. Anita is at astrology@earthlink.net ). Get a free natal horoscope "my money/future life" reading now + copy horoscope as a Gif file graphic! No smarter, more accurate career reading out there! PS. Anita sez, 'get a website for 5$ a month and post articles that YOU LIKE, including all of hers, free, TOO! Be someone who forwards the action, the evolution. LIFE on EARTH IS getting better and it's ranting progressives who did it and now the INTERNET is doing it! JOIN IN!
<=== BACK TO TRACKING THE ECONOMY, an INDEX PAGE
<==BACK TO THE FUTURE
<===BACK TO MONEY SECRETS ONLY THE EXPERTS KNOW
<=== BACK TO SECRETS THE GOV DOES NOT WANT YOU TO KNOW
<===BACK TO THE SECRETS OF THE OLIGARCHS, THE EMPIRE INDEX PAGE
<=== SHOW ME THE FIX INDEX PAGE.
<== SHOW ME THE HAPPY R)EVOLUTION PAGE
<=== BACK TO "GUERILLA CAPITALISM" -- THE SOLUTION!
<==== BACK TO THE "VITAL SIGNS OF A DYING ECONOMY" the "FUTURE" WEBPAGE
<==== BACK TO THE WALL STREET MELTDOWN WEBSITE, with "WHAT TO DO TO SURVIVE" TIPS
<=== BACK TO ENRON PLANET, the DOOMSDAY SCENARIO!
<====BACK TO THE HOLISTIC GOURMET, BON MARCHE
<=== BACK TO THE FRUGAL ARTISAN IMPORT/ EXPORT TYCOON AND HIS "STUFF"
<=== BACK TO THE GLEENERS PAGE
<=== HOW EVEN A POOR PERSON CAN BUY REAL ESTATE
<== BACK TO THE FUTURE INDEX PAGE
<=== BACK TO THE GENTLEMAN FARMER'S GARDEN INDEX
<=== INVESTIGATE DOING DRIP IRRIGATE- LINES in DROUGHT STATES, EZ to learn in ONE DAY, LOW COST STARTUP.
<=== BACK TO "DONE WELL, ACTIVISM IS A DELIGHT and IT CREATES CAREERS for YOU as well as PLANETARY EVOLUTION"
<=== BACK TO THE MALTHUSIAN INDEX PAGE
<===BACK TO THE PHILOSOPHY INDEX PAGE
<=== BACK TO THE LUCK IN LOVE WEBSITE
<== BACK TO THE PROPAGANDA STUDIES WEBSITE, HOW GOV LIES TO YOU
<=== BACK TO ALL POINTS OF THE COMPASS POLITICAL PHILOSOPHY TUTORIAL
<=== BACK TO JERRY'S REFRIGERATOR & the Tin Foil Hat Collection of CONSPIRACY THEORIES!
<====BACK TO THE FREE MONEY WEBSITE
<==== BACK TO THE HARD TIMES WEBSITE
<===== BACK TO THE POVERTY INDEX PAGE
<==== BACK TO THE "TIPS to SURVIVE THE FUTURE" INDEX PAGE
BACK TO THE "FIX YOUR FLAGGING AMBITION" SEMINAR
<== BACK TO THE SNOOKERED INDEX PAGE
<===BACK TO THE REALITY 101 SEMINAR FOR TEENS
<==== BACK TO THE SHOW BUSINESS ARCHIVE for directors writers, video makers, producers and video documentarians
<====HOW TO TINKER WITH OTHER PEOPLE's BRAINS for $ ( TANTRUM YOGA)
<====BACK TO THE NEW AGE EMPOWERING INDEX
<===BACK TO "HOW TO MERCHANDISE YOURSELF" INDEX
<===BACK TO THE ALL ABOUT TAXES INDEX PAGE
<=== MEET ANITA SANDS HERNANDEZ WHO WRITES ALL THESE ARTICLES
<=== TAKE ME TO THE HOLISTIC PET, HOW NEVER TO NEED a VET
<===== TAKE ME TO THE SALMONELLA TIMES, HOW TO SAVE THE CHICKEN
<=== BACK TO THE TRUTHS ABOUT DENTISTRY THAT WILL SAVE YOU 100 THOUSAND!
<=== BACK TO THE SNOOKERED WEBPAGE, HOAXES WE HAVE ALL KNOWN
<==== PORTAL TO 7,000 FILES YOU CAN REPUBLISH FOR FREE, ATTRACT AD REVENUE off a 5$ a month WEBSITE
<===BACK TO EVERYTHING YOU NEED TO KNOW ABOUT WEB STUFF (for dummies)
<===BEAUTIES, HOW TO MARRY A BILLIONAIRE AND SAVE THE WORLD
<=== BACK TO HOW THE POOR MAN CAN BUY REAL ESTATE SEMINAR
<=== BACK TO THE PORTAL to 7,000 LIFE 101 ARTICLES,ALL FREE TO U TO REPUBLISH, (5$ a month WEBSITE will THEN ATTRACT AD REVENUE.)